
A recent study from North Carolina State University experimented using 3D bioprinting to produce plant cells in order to assess the use of the method in biological research. They produced Arabidopsis- (thale cress; often used in genetic research) and soybean cells. Their 3D-printed cells had adequate viability and went on to produce microcalli (groups of plant tissue), indicating that they were successful productions. These findings suggest that 3D bioprinting could be at least as effective as traditional methods for plant cell reproduction.
Methods
Firstly, let’s look at the method used to conduct the experiment: pneumatic extrusion. This is a type of 3D bioprinting in which the researcher utilizes air pressure to force out the biological material through a nozzle. This printed both the bioink and the support structures, likened to scaffolding. For the latter, they tested two different types- agarose and sodium alginate, to assess which one proved more effective. The soy bean cells showed good viability of almost 49% two weeks after creation, which indicates that bioprinting could produce cells with equal viability to traditional manual pipetting. After 5 days, viability within the 3D bioprinted structure was almost 25% for the Arabidopsis cells with agarose ‘scaffolding’.
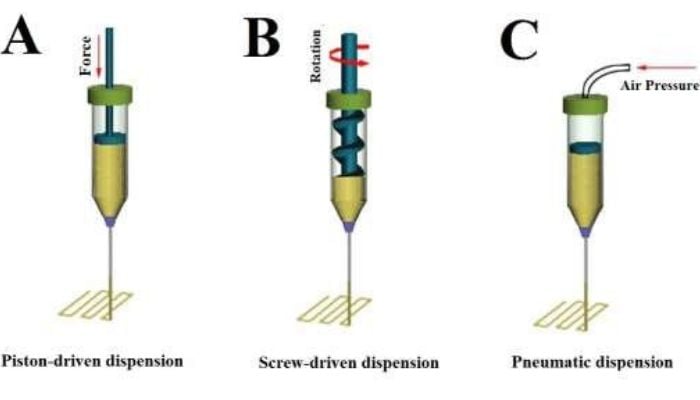
The team used a bioprinting method called pneumatic extrusion or dispension – C. Image credit: Advanced Nanobiomed Research on ResearchGate
For the viable cells, researchers were able to assess production of microcalli (groups of unorganized parenchyma cells, a type of plant cell). The cell cycle reentry coincided with the induction of core cell cycle genes and genes related to cell regeneration. Essentially, they caused the bioprinted cells to restart their cell cycles and reproduce. They found that after 14 days, on average 90% of bioprinted constructs formed 5-6 microcalli. This indicates that 3D bioprinted cells have the potential to undergo cell division and behave as cells produced by traditional methods.
Lastly, scientists examined the cellular identity of bioprinted root plant cells and the microcalli which they had formed. They used markers to track the protein expression and found that cell identity appeared to change over time. They observed that the cells induced genes responsible for the initiation or maintenance of stem cell identity within 3 days of observation. Researchers also tested whether the environment could be changed to evaluate cellular responses by applying high salinity. They found that the cells were less viable under these stress conditions, both meristematic (non-specialised plant cells which can become any type of cell) and differentiated.

Biological research could be improved with the use of 3D Bioprinting.
Results and implications
These results have intriguing implications for 3D bioprinting in biological studies of plant cells. The reasonable viability of cells produced by this method shows that scientists could use it as an alternative to pipetting, in particular as it ‘provides better opportunity for high throughput processing and control over the architecture of the cells after bioprinting’, according to corresponding co-author Professor Ross Sozzani. The production of microcalli indicates that these cells can rejoin the cell cycle and divide as normal cells would. Finally, the study into cell identity indicated that:
“Bioprinted cells can take on the identity of stem cells; they divide and grow and express specific genes,” Van den Broeck said. “When you bioprint, you print a whole population of cell types. We were able to examine the genes expressed by individual cells after 3D bioprinting to understand any changes in cell identity.”
The researchers suggested that further studies could use 3D bioprinting to test cell viability, division and identity in a ‘tunable environment’. 3D bioprinting has previously been used in biological projects such as medicinal organ growth and the production of meat alternatives. This is a very interesting field and is important for scientific discovery not simply business applications, unlike some uses of 3D printing. If you wish to read the full report, it is available on Science.org (log-in required). You can find a summary on the University’s news blog HERE.
*Cover photo credit: Lisa Van den Broeck, NC State University



